I say “free” but in reality most protons in a water-based (aqueous) solution quickly latch onto a water molecule to become a hydronium ion (H₃O⁺). That’s what pH is a measure of, acidity, which refers to how many free protons (H⁺) are hanging out in a solution. Bottom line – don’t change anything you aren’t interested in – this is what we refer to as “controlling variables.” A variable we commonly control is pH, and we keep it in a narrow range using chemicals called “buffering agents.”īy acting as both an acid ( proton-giver) and a base (proton-taker), buffering agents like Tris, sodium phosphate, HEPES, CHAPS, & MOPS, are able to maintain a steady pH so that we keep our molecules happy and don’t have to worry about fluctuations in how many “free” protons (H⁺) are floating around. Just like (unless you’re looking for effects of temperature) you wouldn’t want to compare a reaction performed in a cold room to one performed at “room temp,” you don’t want to compare reaction performed at different pHs (unless that’s the only variable you’ve changed because you’re looking for effects of pH). Basically (no pun intended (this time) – honestly!) every time you want to see how molecules act and interact, you have to make sure that they’re in an okay environment, and you want to make sure that environment stays consistent – especially if you’re trying to compare things. Since I do a lot of protein purifications, I need to make a lot of buffers – and it’s not just for protein purifying that buffer-making is required. 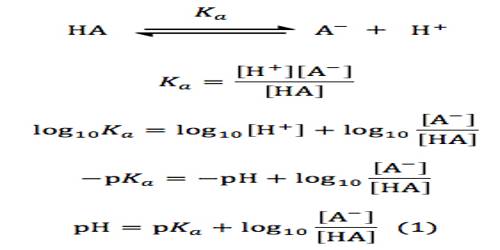
Instead, you usually make up baths for them that resemble their native homes (like the inside of cells) but with minimum molecular “clutter.” While it’s easy to focus on the protein we put in the buffer and forget everything else, the buffer itself matters too! A lot! And it’s precisely because of its buffer-ness that we’re able to “ignore it.” A lot of the buffers I make are made to serve as nice soothing baths for the proteins I’m working with – and just like you wouldn’t want to take a bath in lye (super basic (high pH)) or vinegar (super acidic (low pH)), you shouldn’t ask the proteins and other molecules you’re working with to do so! And you certainly shouldn’t expect them to act the same in those different conditions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
